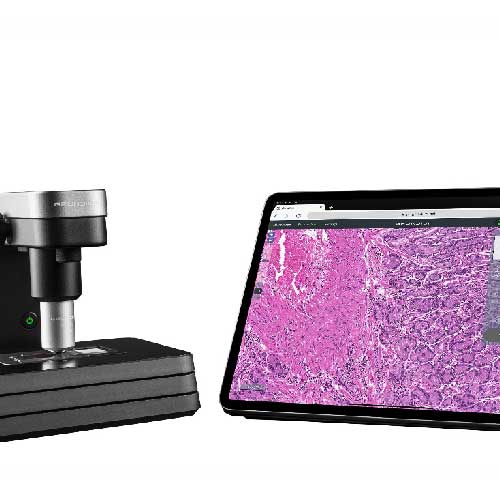
BioSpot Arc的优势:
• 一台设备可同时配置皮升、纳升分液模块
• 搭配TopView Camera成像模块,可对分液过程严格质控
• 独特的体积优化和校准模式,手动输入目标液体体积
• SmartDrop自动校准相机系统,基于成像的液滴体积自动快速校准
• 移液精度CV< 1-3%
• 分液和点样速度快,具备飞行模式
应用方向:
- 芯片研发生产
- 微流控芯片点样
- PCR反应体系setup
- 药物筛选
- 针头药物包被
- 质谱样品制备
- 合成生物学
- 材料测试(燃料电池等)
多媒体资源
新闻 & 事件
申请Demo或询价
Thank you for contacting us!
{{ successMessage }}